 Deep Brain Stimulation (DBS) is a treatment using Activa® Therapy, a brain stimulation technology developed by Medtronic Neurological Therapy Development Group to deliver carefully controlled electrical pulses to precisely targeted areas of the brain involved in movement control.
Deep Brain Stimulation (DBS) is a treatment using Activa® Therapy, a brain stimulation technology developed by Medtronic Neurological Therapy Development Group to deliver carefully controlled electrical pulses to precisely targeted areas of the brain involved in movement control.
It involves placing electrodes, thin flexible wires, through the skull into the deep portions of the brain. The device is similar to a pacemaker; it is to the brain what a pacemaker is for the heart. An external battery is placed subcutaneously with wires that are not visible through skin that then enter the skull to reach deep sections of the brain, usually in one of the regions of the basal ganglia (nerve cell centers deep on each side of the brain from which it is believed dystonia arises.)
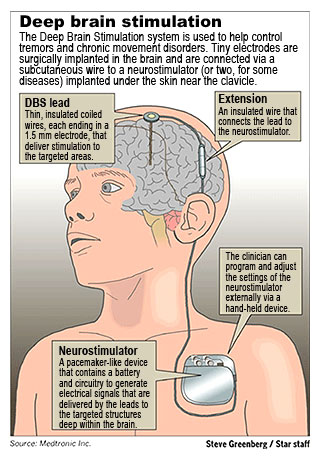 After the surgery adjustment need to be made to the frequency and the power of the electrical stimulus being presented to the nerve cells through the device. If the right adjustment is made as to the rate of delivery and the strength of delivery, remarkable improvement can be seen.
After the surgery adjustment need to be made to the frequency and the power of the electrical stimulus being presented to the nerve cells through the device. If the right adjustment is made as to the rate of delivery and the strength of delivery, remarkable improvement can be seen.
Deep brain stimulation has been given a Humanitarian Device Exemption by the FDA for the treatment of dystonia in 2003.
More and more people with serve cases of cervical dystonia are choosing this option. You need to remember this is a treatment and not a cure. It is not a quick fix surgery. The treatment does not end after surgery.
A person would need to be programmed regularly by a trained neurologist every few months.

